Carbon electron configuration number7/1/2023  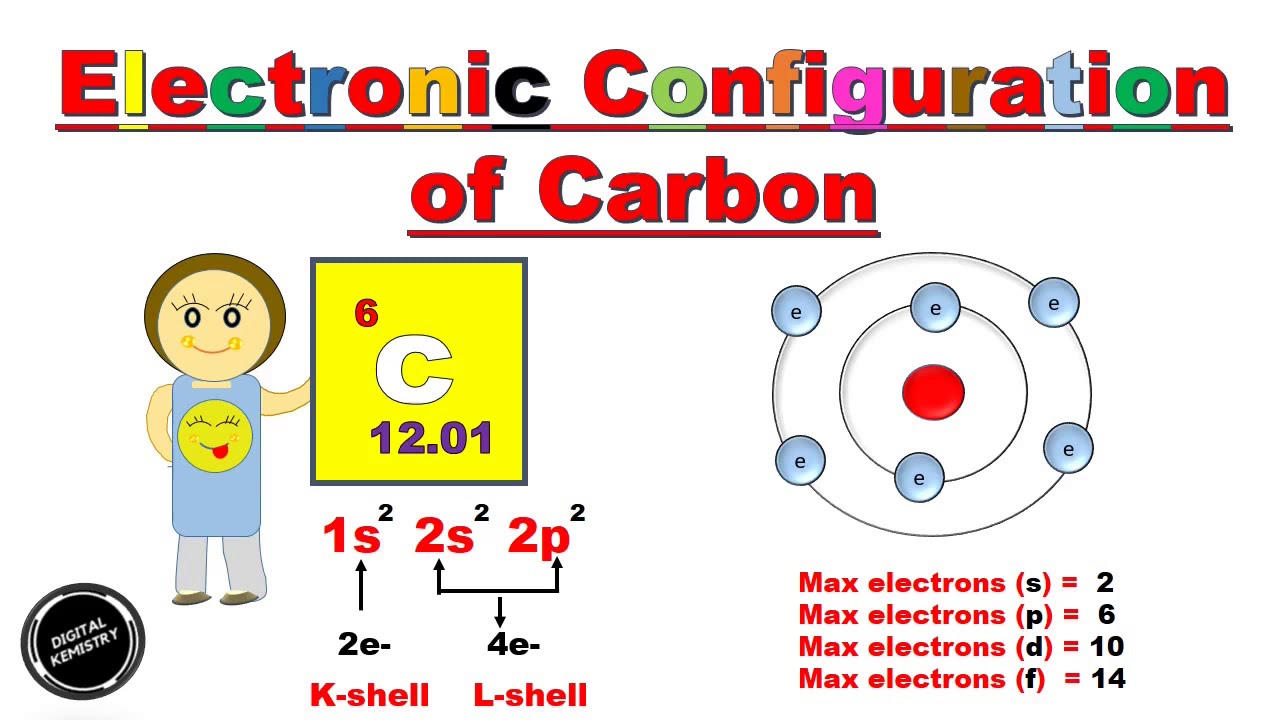
The n = 1 shell is filled with two electrons and three electrons will occupy the n = 2 shell. The fourth electron fills the remaining space in the 2 s orbital.Īn atom of boron (atomic number 5) contains five electrons. Thus, the electron configuration and orbital diagram of lithium are:Īn atom of the alkaline earth metal beryllium, with an atomic number of 4, contains four protons in the nucleus and four electrons surrounding the nucleus. The 18 core electrons are in shells 1, 2, and 3 (1s 2 2s 2 2p 6 3s 2 3p 6).\) ). The outermost and highest energy level is 4, containing two valence electrons (4s 2). The Atomic Number(Z) of Calcium (Ca) is 20, and the electronic configuration of Ca is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2. The ten core electrons are in shells 1 and 2 (1s 2 2s 2 2p 6). The outermost shell is 3 with two valence electrons (3s 2). The Atomic Number(Z) of Magnesium (Mg) is 12, and the electronic configuration is 1s 2 2s 2 2p 6 3s 2. Both have the same number of two valence electrons. Magnesium and Calcium belong to Group number 2. The outermost shell is 3 with 6 valence electrons (3s 2 3p 4). The electronic configuration of S is 1s 2 2s 2 2p 6 3s 2 3p 4. The Atomic Number(Z) of Sulfur (S) is 16 with 16 electrons. The two core electrons are present in shell 1 (1s 2). The farthest shell from the nucleus is 2 with six valence electrons (2s 2 2p 4). The electronic configuration of O is 1s 2 2s 2 2p 4. The Atomic Number(Z) of Oxygen (O) is 8, corelating to 8 electrons. Oxygen and Sulfur belong to the same group 16 in the periodic table and therefore have the same number of 6 valence electrons. The outermost shell is 3, containing five valence electrons (3s 2 3p 3), and the core electrons are in the inner 1 and 2 shells (1s 2 2s 2 2p 6). The electronic configuration of P is 1s 2 2s 2 2p 6 3s 2 3p 3. 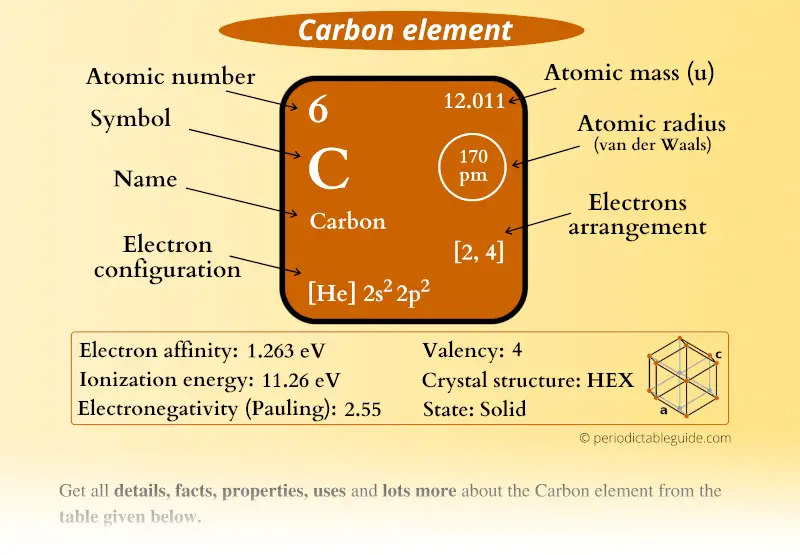 
The Atomic Number (Z) of Phosphorus (N) is 15, with 15 electrons distributed into various shells and orbitals. So, the outermost shell is 2, containing five valence electrons. The Atomic Number (Z) of Nitrogen (N) is 7, and its electronic configuration is 1s 2 2s 2 2p 3. The valence electrons of both Nitrogen and Phosphorus are 5. The elements belonging to the same group have the same valence electrons. The outermost shell 3 contains four valence electrons (3s 2 3p 2), and the remaining are Silicon's core electrons in shells 1 and 2 (1s 2 2s 2 2p 6).Ĭarbon and Silicon belong to the same group 15 in the periodic table. Its electronic configuration is 1s 2 2s 2 2p 6 3s 2 3p 2. The outermost shell is 2, containing four valence electrons (2s 2 2p 2), and the two core electrons are in shell 1 (1s 2). Its electronic configuration after dividing the electrons into shells and orbitals is - 1s 2 2s 2 2p 2. 
So, both Carbon and Silicon will have four valence electrons. The only difference will be their shell number due to the increase in atomic size. All elements belonging to the same group in the periodic table will have the same number of valence electrons (table A). The group number and atomic number is used to determine the valence electrons of an element.Ĭarbon and Silicon belong to the same group 14 in the periodic table. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
